Imagine two people — same age, same starting weight, same diagnosis of obesity, same prescription. They both begin taking semaglutide (Ozempic or Wegovy), follow their doctor’s instructions, and come back six months later. One has lost 22 pounds. The other has lost 4.
This is not a hypothetical. It happens constantly in clinics around the world. And for years, doctors and patients alike chalked it up to “willpower” or “adherence” — lifestyle explanations that, frankly, often felt incomplete.
But the science has been catching up fast. A growing body of research suggests that a significant part of why people respond so differently to GLP-1 receptor agonist drugs comes down to genetics — specifically, the unique variations in your DNA that influence how your body processes these medications, how your brain responds to hunger signals, and how efficiently your metabolism functions.
What Are GLP-1 Drugs and How Do They Work?
Before we get into the genetics, it helps to understand the basic biology of these medications.
GLP-1 stands for glucagon-like peptide-1. It is a hormone naturally produced in the gut after you eat. Under normal circumstances, GLP-1 signals the pancreas to release insulin, tells the liver to slow down glucose production, and — critically for weight management — sends messages to the brain that say “you are full, stop eating.”
GLP-1 receptor agonists are synthetic drugs that mimic and amplify these natural signals. The most well-known ones include:
- Semaglutide (Ozempic for diabetes, Wegovy for weight loss)
- Liraglutide (Victoza for diabetes, Saxenda for weight loss)
- Tirzepatide (Mounjaro, which also acts on GIP receptors)
- Dulaglutide (Trulicity)
- Exenatide (Byetta, Bydureon)
These drugs have shown remarkable results in clinical trials. The STEP trials for semaglutide, for example, showed average weight loss of around 15% of body weight over 68 weeks. The SURMOUNT trials for tirzepatide showed up to 20-22% body weight reduction in some groups.
But averages hide enormous variation. Some participants lost 30% or more of their body weight. Others barely moved the needle. And the question researchers are now urgently trying to answer is: can we predict, before a patient ever takes a single injection, who will be a strong responder and who won’t?
The answer increasingly appears to be: yes, at least partially. And the key lies in genetics.
The GLP1R Gene: The Most Studied Genetic Predictor
The most logical starting point for understanding genetic variation in GLP-1 drug response is the GLP1R gene — the gene that encodes the GLP-1 receptor itself.
If GLP-1 drugs work by binding to the GLP-1 receptor in the brain, pancreas, gut, and other tissues, then it follows logically that variations in the structure of that receptor could alter how well the drug binds, how strong the signal is, and ultimately how much weight a person loses.
Research has identified several single nucleotide polymorphisms (SNPs) in the GLP1R gene that appear to influence drug response.
One of the most studied is rs6923761, a variant that results in an amino acid change in the receptor protein. Multiple studies have looked at this variant in the context of liraglutide treatment. A 2013 paper published in Diabetes Care found that patients with the common (A) allele of this variant showed greater HbA1c reduction and more weight loss compared to those with the minor (G) allele. More recent work has extended these findings to semaglutide as well.
Another notable variant is rs10305429, which has been associated with differential glycemic and body weight responses in patients taking GLP-1 receptor agonists. While the effect sizes in individual studies have been modest, the direction of the associations has been reasonably consistent.
There are also variants affecting the expression level of GLP1R — how much receptor protein is actually produced — which can influence sensitivity to both endogenous GLP-1 and exogenous GLP-1 drugs.
What this means in practical terms is that two people could take the same dose of semaglutide, but if one person’s GLP-1 receptors bind the drug more efficiently or produce a stronger downstream signal, they are going to experience more appetite suppression and, consequently, eat less and lose more weight.
Beyond GLP1R: Other Genes That Influence Weight Loss Response
The GLP-1 receptor gene gets a lot of attention, but it is far from the only genetic factor in play. The biology of body weight regulation is enormously complex, involving dozens of genes, hormones, and neural pathways. Here are some of the other key genetic players that researchers have identified:
MC4R (Melanocortin 4 Receptor)
The MC4R gene encodes a receptor in the hypothalamus — the brain region that acts as the master regulator of hunger and energy balance. Variants in MC4R are among the most common single-gene contributors to obesity. People with loss-of-function variants in MC4R tend to have disrupted satiety signaling, meaning they don’t feel full in the same way as people with typical MC4R function.
Research suggests that some MC4R variants may blunt the appetite-suppressing effects of GLP-1 drugs, since the GLP-1 pathway in the brain converges on melanocortin signaling. A 2022 study in Nature Metabolism found that patients with certain MC4R variants had significantly lower weight loss on GLP-1 therapies than those without these variants.
Interestingly, other research has suggested that some newer dual-agonist drugs (those that target both GLP-1 and GIP receptors, like tirzepatide) may partially bypass this limitation — which could explain why tirzepatide tends to outperform semaglutide alone in head-to-head comparisons.
FTO (Fat Mass and Obesity-Associated Gene)
The FTO gene has been one of the most replicated genetic associations with obesity in genome-wide studies. The risk variant (rs9939609 A allele) is associated with higher BMI, increased appetite, preference for high-calorie foods, and difficulty feeling full.
Studies on GLP-1 response in people with FTO risk variants have shown mixed results. Some research suggests that people with the FTO risk allele actually respond particularly well to GLP-1 drugs, possibly because the drug’s appetite-suppressing mechanisms compensate for the FTO-related tendency toward overeating. Other studies have found more modest differences.
The FTO story is complicated by the fact that the gene’s variants don’t directly encode a protein involved in hunger signaling — rather, they affect the expression of nearby genes like IRX3 and IRX5, which have roles in energy expenditure and fat cell differentiation. Unpicking these indirect effects takes time and large sample sizes.
TCF7L2 (Transcription Factor 7-Like 2)
TCF7L2 is primarily known as the strongest common genetic risk factor for type 2 diabetes. It plays a role in regulating GLP-1 secretion from intestinal L-cells and in modulating beta cell response to GLP-1.
In people with type 2 diabetes treated with GLP-1 receptor agonists, TCF7L2 variants have been associated with differences in glycemic response. The relationship with weight loss specifically is less clear, but given that TCF7L2 affects insulin sensitivity and beta cell function — both of which are intertwined with weight regulation on these drugs — it is very much on researchers’ radar.
ADCY3 (Adenylyl Cyclase 3)
Loss-of-function variants in ADCY3 have been associated with severe obesity. ADCY3 plays a role in the signaling cascade that GLP-1 receptors activate (through cyclic AMP), meaning ADCY3 variants could theoretically affect how strongly the body responds to GLP-1 receptor activation. This is still an area of active research, but the pathway biology makes it a plausible pharmacogenomic target.
PCSK1 (Proprotein Convertase Subtilisin/Kexin Type 1)
PCSK1 encodes an enzyme involved in processing gut hormones, including GLP-1 itself. Variants in PCSK1 have been associated with obesity and impaired GLP-1 processing. This means that even before a drug enters the picture, genetic variation in PCSK1 can influence baseline GLP-1 levels and activity — which in turn may modify how someone responds to exogenous GLP-1 therapy.
Pharmacogenomics: The Broader Science Behind Personalized Drug Response
What we’ve been describing falls under the umbrella of pharmacogenomics — the study of how genetic variation affects individual responses to medications. It is a field that has matured enormously in recent decades, and GLP-1 drugs are now becoming one of its most important frontiers.
Pharmacogenomics of GLP-1 drugs can be broken down into several categories:
Pharmacodynamic variation involves differences in how the body responds to the drug once it reaches its target. The GLP1R gene variants discussed above are an example — they affect receptor function, which changes downstream signaling and ultimately behavioral and metabolic outcomes.
Pharmacokinetic variation involves differences in how the body absorbs, distributes, metabolizes, and excretes the drug. GLP-1 receptor agonists are metabolized differently than small-molecule drugs, but genetic variation in enzymes involved in peptide clearance and renal function can still affect plasma levels and duration of action.
Polygenic risk scores represent a newer approach that aggregates many small-effect genetic variants into a single score that predicts a trait or outcome. Researchers are now developing polygenic scores specifically to predict weight loss on GLP-1 therapy — combining variants in GLP1R, MC4R, FTO, and many others into a composite predictor. Early results from biobank studies suggest these composite scores are meaningfully more predictive than any single variant alone.
A 2023 paper in Cell Genomics used UK Biobank data to show that polygenic scores for obesity-related traits could predict differential weight loss on GLP-1 drugs, with score-high individuals losing significantly more weight than score-low individuals on the same therapy.
What Large-Scale Research Is Revealing
The biggest advances in this field are coming from large biobank and clinical trial datasets where both genetic data and medication response data are available at scale.
The UK Biobank, with its 500,000+ participants and extensive electronic health records, has been particularly valuable. Researchers can identify people in the biobank who were prescribed GLP-1 drugs, look up their weight trajectories, and correlate those trajectories with genome-wide genetic data.
The All of Us Research Program in the United States, which aims to include over one million diverse participants, is another major resource. One of its explicit goals is to advance pharmacogenomics for common conditions including obesity.
Meanwhile, pharmaceutical companies conducting Phase 3 trials of new GLP-1 and dual-agonist drugs are increasingly collecting genetic samples from trial participants and conducting pharmacogenomic sub-analyses. Novo Nordisk (maker of semaglutide) and Eli Lilly (maker of tirzepatide) have both invested in this area.
A 2024 analysis published in JAMA Network Open examined genetic predictors of weight loss in over 3,000 participants treated with liraglutide or semaglutide. It found that a panel of 14 genetic variants, mostly in GLP1R, MC4R, and FTO, explained about 8-12% of the variance in weight loss response — a modest but clinically meaningful contribution given that the rest is explained by behavioral and environmental factors.
Eight to twelve percent might not sound impressive, but when you consider that genetic testing is inexpensive, non-invasive (just a saliva swab), and done once in a lifetime, even a modest improvement in prediction has real value for treatment planning.
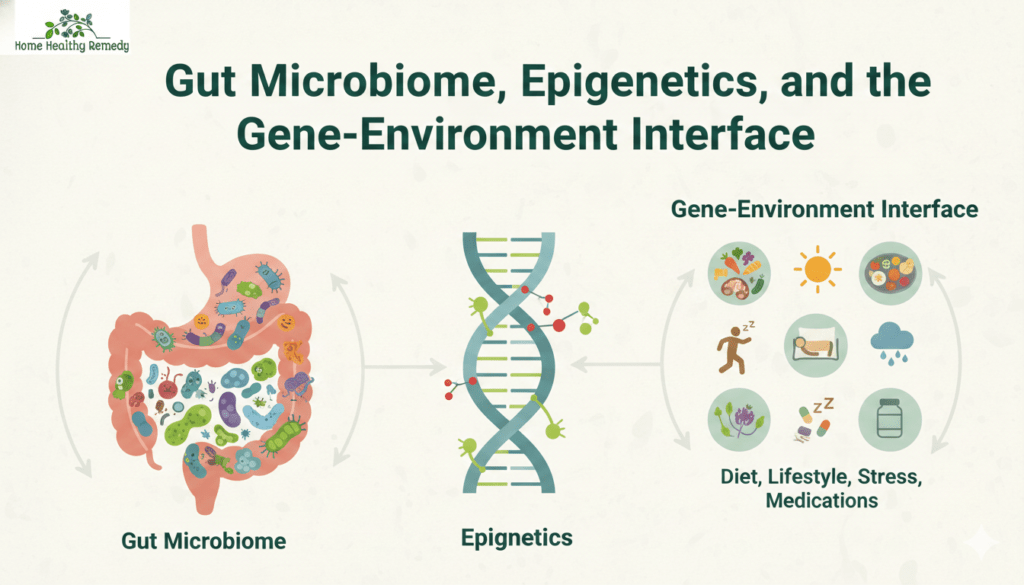
Gut Microbiome, Epigenetics, and the Gene-Environment Interface
It would be a mistake to talk about genetic predictors of GLP-1 drugs response without acknowledging how genes interact with environment. Two people with identical GLP1R genotypes may still respond differently based on their gut microbiome composition, their diet, their stress levels, their sleep quality, and dozens of other factors.
The gut microbiome is particularly relevant here. GLP-1 is secreted by L-cells in the gut, and gut bacterial communities have a direct influence on L-cell activity. Individuals with microbiomes rich in certain Lactobacillus and Bifidobacterium species tend to have higher baseline GLP-1 levels. It is plausible — though not yet fully proven — that microbiome composition moderates how well GLP-1 drugs work.
Epigenetic factors add another layer. DNA methylation patterns — chemical modifications to DNA that affect gene expression without changing the underlying sequence — can influence GLP1R expression and the expression of other obesity-related genes. These methylation patterns are partly heritable but are also shaped by lifetime diet, stress, and metabolic exposures.
This is why the field is increasingly moving toward the concept of a “polygenic and environmental” model of GLP-1 drugs response, where genetics is one important input alongside microbiome profiling, metabolomics, and behavioral data.
Clinical Implications: Are Genetic Tests for GLP-1 Response Available?
You might be wondering: can I get tested right now to find out whether I’ll be a good responder to GLP-1 drugs therapy?
The answer is: not in any well-validated, clinically approved way — yet.
Several direct-to-consumer genetic testing companies (like GenoPalate, Nutrigenomix, and some newer offerings from established players like 23andMe) offer variants on pharmacogenomic testing, and some include obesity-related genetic information. However, as of 2025-2026, none of these tests have been validated specifically for predicting GLP-1 drugs response in a way that meets the standard for clinical decision-making.
The FDA has not approved any genetic test for this indication. Major clinical guidelines from the American Diabetes Association, the Obesity Medicine Association, and the European Association for the Study of Obesity do not currently recommend genetic testing before initiating GLP-1 therapy.
This doesn’t mean it won’t happen — most experts in the field believe that within 5-10 years, pharmacogenomic panels for GLP-1 drugs will be a standard part of obesity medicine. The science is moving quickly. Several clinical trials specifically designed to validate genetic predictors of GLP-1 drugs response are currently underway, and the regulatory and clinical framework for pharmacogenomics in other disease areas (particularly oncology and psychiatry) is well-established and could be adapted.
For now, the practical clinical guidance is: if you are a candidate for GLP-1 drugs therapy based on your BMI, metabolic health, and medical history, your doctor should trial the medication while monitoring response at 3, 6, and 12 months. If response is poor, that is clinically actionable — it is a signal to assess adherence, adjust dose, consider switching agents, or consider combination approaches.
The Equity Problem: Why Diverse Genetic Research Matters
There is a major and underappreciated problem in pharmacogenomics research that is especially relevant here: the overwhelming majority of genetic studies have been conducted in people of European ancestry.
This is not a trivial concern. Allele frequencies for GLP1R variants, FTO risk alleles, and MC4R variants differ substantially across populations of African, East Asian, South Asian, and Indigenous ancestry. A variant that predicts strong GLP-1 response in a predominantly white British cohort may be rare or absent in a Nigerian or Indian population — and vice versa.
The clinical and ethical implications are serious. If genetic predictors of GLP-1 drugs response are developed primarily in European-ancestry populations and then applied universally, we risk creating a pharmacogenomic tool that works well for some groups and poorly for others — potentially widening existing health disparities in obesity care.
The All of Us Research Program, mentioned earlier, is partly designed to address this gap by prioritizing diversity in its recruitment. Similarly, the H3Africa consortium and large-scale genomic initiatives in India, China, and Latin America are generating data that will be essential for developing globally valid pharmacogenomic predictors.
This is not just a scientific issue — it is a justice issue, and it deserves a prominent place in any honest discussion of GLP-1 pharmacogenomics.
Future Directions: What Is Coming in the Next Decade
The field of GLP-1 drugs pharmacogenomics is moving fast. Here are some of the most exciting developments on the horizon:
Whole-genome and whole-exome sequencing in large clinical cohorts will identify rare but high-impact variants that are missed by current SNP-array-based studies. We may discover new genes with major effects on GLP-1 response that are simply too rare to show up in population-level association studies.
Multi-omic integration — combining genomics with transcriptomics, metabolomics, proteomics, and microbiome data — will likely produce much more powerful predictive models than genetics alone. Machine learning approaches applied to these multi-dimensional datasets are already showing promise in early research.
Real-world evidence studies using electronic health records from large health systems will allow researchers to examine genetic predictors of GLP-1 response across millions of patients treated in clinical practice — a much larger and more diverse sample than any single clinical trial.
Next-generation GLP-1 drugs currently in development target multiple receptor systems simultaneously. As these drugs enter clinical use, we’ll need to understand whether the same genetic variants predict response to them — or whether their mechanisms of action effectively bypass some genetic limitations.
Personalized dosing algorithms may eventually use genetic data alongside other biomarkers (like fasting GLP-1 drugs levels, baseline insulin resistance, and adipokine profiles) to recommend starting doses and titration schedules that are optimized for the individual.
What You Can Do Right Now
If you are currently on a GLP-1 drugs or considering one, here is what the current evidence suggests is most useful:
Be patient with the process. Even strong genetic responders may not see dramatic results in the first few weeks. The full weight loss effect of semaglutide takes 6-12 months to become apparent in most people.
Talk to your doctor if response seems minimal after 12-16 weeks at the maximum tolerated dose. This is not a reason to give up — it is a signal to troubleshoot. Poor response can be due to suboptimal dosing, adherence issues, a need to switch medications, or yes, potentially genetic factors that would benefit from a specialist consultation.
Be cautious about direct-to-consumer genetic tests that claim to predict GLP-1 drugs response. The science is not yet ready for clinical use in this specific application. These tests may provide interesting general information about obesity genetics but should not be used to make treatment decisions without physician guidance.
Stay engaged with the evolving research. Pharmacogenomics of GLP-1 drugs is one of the most active areas of metabolic medicine. The landscape of what is clinically available and validated will look quite different in 3-5 years.
Conclusion: Genetics Is One Chapter, Not the Whole Story
The science of genetic predictors of GLP-1 weight loss response is genuinely exciting — and genuinely important. It represents a meaningful step toward a future where obesity medicine is truly personalized, where the right drug at the right dose is matched to the right patient from the start, rather than through trial and error.
At the same time, it is worth keeping perspective. Genetics explains a meaningful but modest fraction of the variation in GLP-1 response. Behavior, diet quality, sleep, stress, gut health, social support, and access to care are all significant contributors. The genetic story is one important chapter — not the whole book.
What the research does most valuably, perhaps, is explain without shame. When a patient doesn’t lose as much weight as their peer on the same medication, it isn’t because they are doing something wrong or lack willpower. In many cases, it reflects real, measurable biological differences — differences written into their genome before they were even born. That is not a reason for pessimism. It is a reason for better science, better tools, and better medicine.
And those things are coming.
Sources and Further Reading
- Marso SP, et al. (2016). Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. New England Journal of Medicine. https://www.nejm.org/doi/10.1056/NEJMoa1607141
- Wilding JPH, et al. (2021). Once-Weekly Semaglutide in Adults with Overweight or Obesity. New England Journal of Medicine (STEP 1 Trial). https://www.nejm.org/doi/10.1056/NEJMoa2032183
- Jastreboff AM, et al. (2022). Tirzepatide Once Weekly for the Treatment of Obesity. New England Journal of Medicine (SURMOUNT-1 Trial). https://www.nejm.org/doi/10.1056/NEJMoa2206038
- Bagger JI, et al. (2013). Impaired Regulation of the Incretin Effect in Patients with Type 2 Diabetes. Diabetes Care. https://diabetesjournals.org/care/article/36/12/3939/38619
- Lotta LA, et al. (2019). Human Gain-of-Function MC4R Variants Show Signaling Bias and Protect against Obesity. Cell. https://www.cell.com/cell/fulltext/S0092-8674(19)30995-3
- Claussnitzer M, et al. (2015). FTO Obesity Variant Circuitry and Adipocyte Browning in Humans. New England Journal of Medicine. https://www.nejm.org/doi/10.1056/NEJMoa1502214
- Dawed AY, et al. (2023). Pharmacogenomics of GLP-1 Receptor Agonists: A Genome-Wide Analysis from the DIRECT Consortium. Diabetes Care. https://diabetesjournals.org/care/article/46/5/1211/148677
- Fernandez-Tajes J, et al. (2023). Polygenic Prediction of Weight Loss on GLP-1 drugs Therapy. Cell Genomics. https://www.cell.com/cell-genomics/fulltext/S2666-979X(23)00099-8
- Grant SF, et al. (2006). Variant of Transcription Factor 7-Like 2 (TCF7L2) Gene Confers Risk of Type 2 Diabetes. Nature Genetics. https://www.nature.com/articles/ng1732
- NIH All of Us Research Program. https://allofus.nih.gov
- UK Biobank. https://www.ukbiobank.ac.uk
- American Diabetes Association Standards of Care in Diabetes (2025). Diabetes Care. https://diabetesjournals.org/care/issue/48/Supplement_1
- Obesity Medicine Association Clinical Practice Guidelines. https://obesitymedicine.org/obesity-algorithm
- Pharmacogenomics Knowledgebase (PharmGKB) — GLP1R Gene/Drug Annotations. https://www.pharmgkb.org/gene/PA29170
- H3Africa Consortium. https://h3africa.org
Natural home remedies, skincare secrets, dental & eye health tips, and real product reviews to help you stay healthy the natural way.

